Granting Exceptional Reimbursement under Section 16
The Public Health Insurance Act No. 48/1997 Coll. provides in Section 16:
“The competent health insurance fund shall reimburse, pursuant to Section 19(1)(a), in exceptional cases healthcare services otherwise not covered by health insurance, provided that the provision of such healthcare services constitutes the only option in view of the insured person’s health condition.”
That such requests are not rare is evidenced, for example, by statistics published by the General Health Insurance Company of the Czech Republic.
Number of applications submitted to VZP CR under Section 16
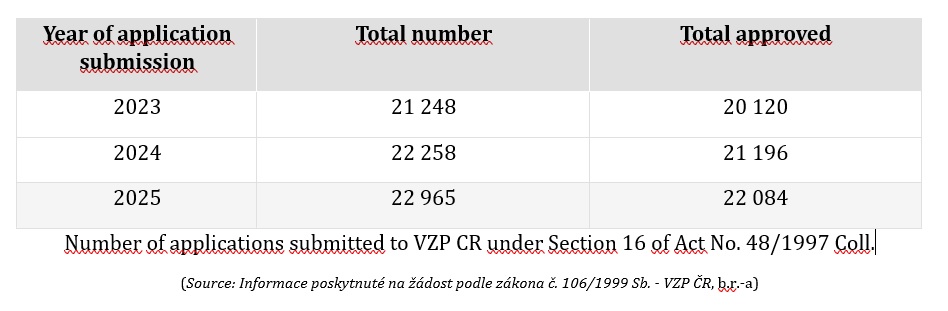
The assessment itself is not governed by any formal methodology. Reviewing physicians of Czech health insurance funds assess each case individually and, based on their professional experience, scientific literature and recommendations, in conjunction with the specific health condition of the insured person, determine whether the above statutory criteria have been met (Information provided pursuant to Act No. 106/1999 Coll. – VZP ČR).
The assessment also relies on established judicial practice, as courts have repeatedly and consistently interpreted the conditions set out in Section 16.
Judicial Approach to the Assessment Criteria
In each case, it should be assessed whether the medicinal product is effective, whether no alternative treatment exists, and whether the case is exceptional.
Effective medicinal product
The mere marketing authorisation of a medicinal product does not automatically establish entitlement to reimbursement under Section 16. However, it should be taken into account that the marketing authorisation process includes, inter alia, an assessment of the product’s effects on patients’ health and a determination that its benefits outweigh potential risks.
Only therapeutic option
It is essential to assess whether, according to the current state of medical knowledge and available evidence, the medicinal product represents the only option offering a reasonable prospect of improving the patient’s health condition.
Fulfilment of this criterion is partly linked to the formal authorisation of the product, but primarily to the recommendation of the treating physician, who assesses the treatment as the only realistic therapeutic option for the specific patient, including from the perspective of safety.
Exceptional nature of the case
The exceptional nature of the case cannot be assessed solely based on the patient’s health condition; other circumstances must also be taken into account. This requires a comprehensive evaluation of the patient’s health status, the course of the disease and the individual situation.
All of the above criteria must be assessed individually. It is therefore not possible to generalise the appropriateness of treatment; each application must be assessed on a case-by-case basis, taking into account the specific patient’s condition.
Within judicial proceedings, it has also been acknowledged that, in certain cases, reimbursement under Section 16 may be claimed not only prospectively but also retrospectively, for example in the form of reimbursement of costs previously paid by the patient, including in cases involving the patient’s successors.
In general, however, reimbursement under Section 16 should always be applied as an exceptional mechanism, not as a systemic or routine solution.
Sources referenced in the article:
Aktuální stav—infoDeska Městský soud v Praze. (b.r.). Získáno 25. březen 2026, z https://infodeska.justice.cz/subjekt.aspx?subjkod=201000&s=a#Ca
Informace poskytnuté na žádosti podle zákona č. 106/1999 Sb. - VZP ČR. (b.r.-a). Získáno 25. březen 2026, z https://www.vzp.cz/o-nas/informace/odpovedi-na-zadosti-o-informace
Informace poskytnuté na žádosti podle zákona č. 106/1999 Sb. - VZP ČR. (b.r.-b). Získáno 25. březen 2026, z https://www.vzp.cz/o-nas/informace/odpovedi-na-zadosti-o-informace?rok=2025
NSS Hledání—Vyhledávač NSS. (b.r.). Získáno 25. březen 2026, z https://vyhledavac.nssoud.cz/Home/Index?formular=4
Are you interested in reading regular commentaries on decisions by Pharmeca a.s.? Feel free to contact us.
At Pharmeca, we help you navigate the complex landscape of pharmaceutical and medical device information. We also offer flexible services that can be tailored to your needs at any time.
Our market position and experience allow us to support you whenever you need expert guidance.
Our knowledge, your opportunity.
The text was
translated using ChatGPT 5.3.

