Orphan Medicinal Products from the Perspective of Decision-Making Practice
In 2025, 12 decisions of the State Institute for Drug Control (SÚKL) became legally effective in administrative proceedings relating to the determination of price and reimbursement for orphan medicinal products. Although the system introduced in 2022 is formally structured around SÚKL decisions, the Institute may not deviate from the binding opinion of the Ministry of Health of the Czech Republic.
A simplified process scheme follows:
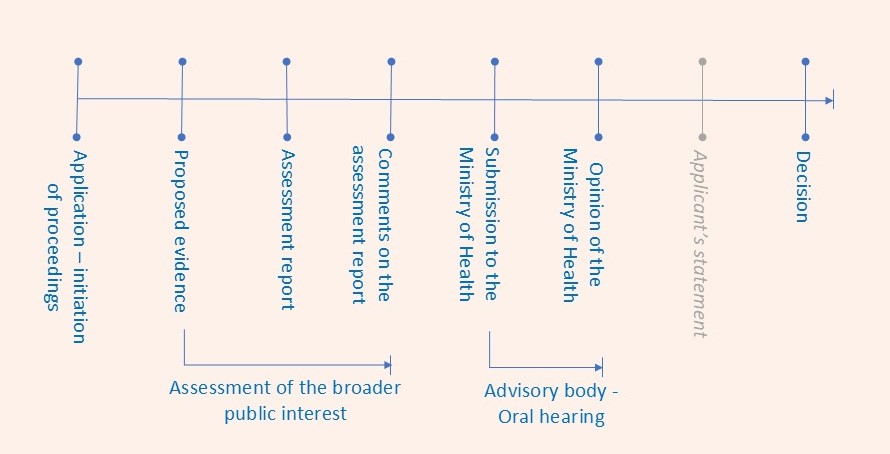
During the past year, the Ministry of Health agreed in eight cases with the determination of reimbursement at the level and under the conditions proposed in SÚKL’s assessment report. In four cases, however, it did not approve the proposed reimbursement.
Among the reasons for issuing a negative opinion were the results of pharmacoeconomic evaluations indicating a significant budget impact on the public health insurance system that would be contrary to the public interest. According to the Ministry’s opinions, the data submitted in the proceedings did not demonstrate sufficiently statistically significant and convincing clinical benefits to justify the high expenditure required from the healthcare system.
Additional reasons cited by the Ministry for refusing reimbursement included an unacceptable assessment of therapeutic efficacy and safety, where the clinical studies presented lacked sufficient data and relevant parameters to confirm efficacy, safety and treatment effectiveness. At the same time, real-world evidence was missing to demonstrate that the effect observed in clinical trials would be durable and transferable into routine clinical practice.
In the above-mentioned cases, the Ministry’s opinions were aligned with the recommendation of its advisory body. In one case, however, the Ministry issued a negative opinion despite the advisory body recommending approval of reimbursement subject to stricter indication criteria. The Ministry justified this step by stating that the observed prolongation of survival was only indicative, and that the impact of the medicinal product on overall survival or on delaying transplantation was uncertain and could not be specified with sufficient precision. In this situation, the Ministry considered it more appropriate to maintain the possibility of individual reimbursement under Section 16 of the Public Health Insurance Act.
Are you interested in reading regular commentaries on decisions by Pharmeca a.s.? Feel free to contact us.
At Pharmeca, we help you navigate the complex landscape of pharmaceutical and medical device information. We also offer flexible services that can be tailored to your needs at any time.
Our market position and experience allow us to support you whenever you need expert guidance.
Our knowledge, your opportunity.
Article is based on publicly available texts from the decisions of the Ministry of Health of the Czech Republic and the State Institute for Drug Control (SÚKL).
The text was translated using ChatGPT 5.2.

